The aufbau principle and electron configurations
Issue 368 | Page 15 | Published Mar 2018
Description
The use of the aufbau principle to predict the electron configurations of atoms (in their ground states), and therefore explain the layout of the periodic table, is a key concept when teaching atomic structure to pre-university chemistry students.
More from this issue
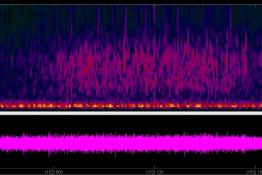
The participation of King Edward VI High School for Girls in the UK School Seismology Project resulted in a research study into the correlation...
Journal Article
Many of my really ‘best’ experiments involve a lot of preparation and often specialised apparatus, but for my ‘top ten’ I have chosen experiments...
Journal Article
This activity describes how students can use acid–base titrimetry to investigate how adding salt to the aqueous phase may change the value of the...
Journal Article